Antimicrobial Protection to Reduce Hospital Associated Infections
Hospital Associated Infections (HAI) are the most common complication of hospital care and one of the top 10 leading causes of death in the USA as reported by The Agency for Healthcare Research and Quality [1]. The most common pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter spp), commonly known as the ESKAPE pathogens, are the leading causes of HAIs. Contracting a HAI can often lead to increased morbidity and can have a devastating effect on physical, mental, and financial health.
In addition to this, HAIs cost the healthcare system billions of dollars a year in added expenditure. Recent research suggests that a growing number of HAIs are caused by pathogens that have become resistant to the antimicrobial medications typically used to control them. [2] The US Center for Disease Control and Prevention identifies that nearly 1.7 million hospitalized patients annually acquire HAIs while being treated for other health issues, and that more than 98,000 patients (1 in 17) die due to these.
This paper presents a protective nano-structured coating with the capability to rapidly kill microbes and pathogens, resulting in a self-sanitizing surface which can be applied to high contact areas throughout hospital and healthcare settings.
The patentend iC-nano™, Infection Control via Nanotechnology, material can be applied to both 2D and 3D surfaces. This work focuses on the technology when applied to observation machines in patient rooms, out-patient check-in kiosks, push plates leading onto various wards and lever door handles throughout the Royal Liverpool University Hospital in the United Kingdom.
For the application on screens, the transparent coating is deposited on flexible polyethylene terephthalate substrates produced in an in-line industrial system. The coating technology could easily be scaled up to a roll-to-roll (R2R) production, to result in a larger throughput of material for commerical use. A large batch coater at Gencoa produced the coatings on the push plates, pull handles and lever door handles.
Introduction
This paper details the use of nanotechnology as a viable solution in the fight against the spread of infection through healthcare settings with the aim of reducing the need for antibiotics and associated antimicrobial resistance (AMR). Throughout a hospital setting, touch surfaces are manually cleaned with chemicals to reduce the microbes and bacteria that are present. This way of sanitation has been shown to be essential in controlling the spread of infections but comes at a high financial cost due to labor as well as the environmental impact of such cleaning agents. The frequency and effectiveness of this type of cleaning is also highly variable, and therefore the aim of this work is to replace this uncertainty with a means to target certain critical touch areas, guaranteeing a consistently 'clean' surface which does not rely upon a human factor. This technology will be in the form of a thin, flexible adhesive pad that is adhered to the touch surface as well as direct coating onto parts. Some of the most common nosocomial pathogens can survive or persist on surfaces for months and therefore be a continuous source of transmission (see table 1).
This is why it is imperative to develop a solution to rapidly kill the ESKAPE(e) pathogens without relying on manual cleaning.
A novel copper-based surface coating has been developed which combines a broad spectrum antimicrobial action with mechanical durability. Unlike other transparent biocidal coatings, this new compound does not require UV light activation, and can therefore be used indoors.
The technology has been applied in strategic locations within the Royal Liverpool University Hospital to study the effect of the antimicrobial surfaces in comparison with current cleaning methods and the potential to reduce Hospital Associated Infections.
The development of the coating and the production of the material by magnetron sputtering in an industrial batch system will be detailed. Focusing on the ESKAPE(e) pathogens, the antimicrobial coated surfaces are closely monitored and regularly sampled to determine if the copper-based material can in fact reduce the transmission of these common HAIs in a hospital environment
Experimental Setup
Cleaning Process & Coating Deposition
A large industrial batch Physical Vapor Deposition (PVD) system was commissioned to produce the coatings on the handles and push plates. The system can house seven 1.4-1.5m length planar magnetrons. However, only two magnetrons were in use for this specific process.
The system was comprised of the main process chamber connected to four turbomolecular pumps with average pumping speeds of 1500 l/s which evacuate the chamber. Once the pressure reached ~5 x 10-5 mbar, a series of heaters were activated to raise the internal temperature of the chamber to ~30°C. The increase of heat aids the outgassing of the substrates and helped reduce the hydrogen and water vapor content present in the system. This stage was closely monitored with the Gencoa gas sensing unit, Optix [6].
The main process chamber is equipped with a Gencoa linear ion source and two Gencoa planar magnetrons (1500mm x 125mm). The target materials were copper-based alloys.
Before depositing the active coating, the substrates were rotated in front of a linear ion source. Substrate cleaning and surface preparation prior to the deposition can significantly improve the quality of the coating and increase the coating adhesion. By pretreating the substrate, the ion source activates the surface and liberates the moisture from the substate. By adding a small amount of O2 during this stage any hydrocarbons are removed before the sputtering process.
To ensure good control and reproducibility of the reactive process, feedback control units were installed on the system. The Optix was used to analyze the reactive gas in the chamber, in this case oxygen (O2) and nitrogen. The Optix can measure the real time partial pressures of the gas composition in the chamber. This is achieved by generating a remote plasma in the head of the unit. A spectrometer housed in the unit looks at the remote plasma and displays the optical emission spectrum through the software in real time. A feedback loop is controlled by pairing the Optix with the Gencoa Speedflo process control unit [7]. The mass flow controller (MFC), which delivers the reactive gas (O2) to the system, was operated by the Speedflo. The MFC was actuated based on a set point defined by the user. This set point is decided based on the amount of excess oxygen in the system. The voltage signal from the Optix, which is proportional to the excess amount of O2, is fed back to the Speedflo, and depending on the set point chosen, the amount of reactive gas being delivered is adjusted. The reactive sputtering feedback control loop maintained the same process conditions throughout the deposition and allowed for a fully reproducible process.
Throughout the development phase, it was found that a lower deposition energy which resulted in a nano textured top surface was preferred. The deposition energy was controlled by limiting the plasma/substrate interaction. By implementing feedback control units, manipulating the gas chemistry, and controlling the deposition energy, we were able to engineer the optimal surface topography which yielded the best antimicrobial results.
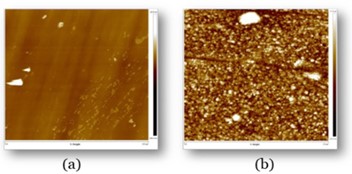
Samples were sent to the University of Liverpool’s Open Innovation Hub for Antimicrobial Surfaces for Atomic Force Microscopy (AFM) analysis. The results shown in Fig. 2 show the significant difference between the uncoated (a) and coated (b) surfaces. The uncoated sample (a) showed a smooth and featureless top surface which yielded poor results when tested for antimicrobial activity. The coated surface (b), however, depicts the nano textured topography which resulted in a LOG6 biocidal effect.
Application
In-situ Testing
Through a government-funded project, granted by Innovate UK (Project Number iUK 10026942), Gencoa worked in partnership with Liverpool University Hospital NHS Foundation Trust, Liverpool School of Tropical Medicine (LSTM) and Diamond Coatings Limited. Access was granted to various locations throughout the Royal Liverpool University Hospital, including several wards and the main atrium. Secondary testing locations were also provided throughout the LSTM Accelerator Building. These secondary locations allowed for constant monitoring and sampling by the biologists working at LSTM during the first phase of testing.
Door panels and handles, similar to those used in the Royal Liverpool Hospital, were coated, and transported to Liverpool School of Tropical Medicine (LSTM). The coated surfaces were installed throughout the school. These were regularly cleaned using the standard cleaning protocol previously in place and a biologist at LSTM sampled both the coated and uncoated surfaces for comparison.
A 10cm2 area on each door panel and handle was swabbed for 30 seconds in a side-to-side motion whilst slowly twirling the swab between the finger and thumb. The swab was then transferred into a stomacher bag containing 10 ml of buffered peptone water (BPW). This was then immediately taken to the microbiology laboratory where it was put in the stomacher with agitation for 1 minute and appropriate dilutions plated out on Mueller-Hinton Agar plates (for total CFU counts) and chromogenic agar to detect specific healthcare-associated pathogens including members of the Enterobacteriaceae. The agar plates were incubated over night at 37°C and colonies enumerated.
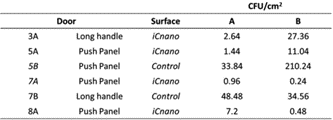
Table 2 details the total CFU/cm2 for various handles and push panels located throughout the school. There is a significant difference between the control and the coated iC-nano surfaces. The uncoated, control surfaces delivered a far higher CFU count per unit area compared to the coated handles.
Results
Antimicrobial Testing
The antimicrobial coatings detailed here work through electrochemistry, and specifically, electrochemistry via oxidative catalysis. The copper ions released from the iC-nano coating can penetrate the bacterial cell wall, oxidizing and changing the cell membrane, causing cell death. In this case, the oxygen for the catalytic oxidation is supplied by the atmosphere. If the growth features of vacuum deposited layers have some degree of ‘separation’ to their neighbors, it will provide a subtle difference in electrochemistry. Once the growth features are bridged by an organic body, it creates a highly effective biocidal mechanism.
Antimicrobial tests were carried out in the laboratories LSTM by inoculating 2cm2 coupons, both uncoated and coated, with 105 and 106 colony forming units (CFUs) of Staphylococcus aureus.
Following a 4-hour incubation at room temperature, the removal of all bacteria cells using a stomacher, dilution and plating out onto suitable agar plates was carried out and the total CFUs enumerated. The reference sample in this case was an uncoated coupon of PET.
Sample A was the uncoated reference and showed the least biocidal activity of the various coatings tested. Sample D (BSM001) was the only coated coupon which did not result in a complete kill of the Staphylococcus aureus. This was expected as it was the only coated sample without any copper content. This reiterated the fact that copper is the active antimicrobial agent. Further testing was carried out on the same samples with the remaining ESKAPE(e) pathogens. This secondary testing saw the samples inoculated with the bacteria for 1 hour as opposed to the primary 4-hour contact time. Apart from sample BSM-001, all samples resulted in a complete kill for all ESKAPE(e) pathogens.
Through this work we have demonstrated that the coatings produced resulted in 100% cell death when incubated between 1 to 4 hours. The International Standard for this type of anti-microbial test is conducted over a 24-hour period However, for the application in a healthcare setting, we needed to prove the efficacy of the coated surfaces over a shorter period.
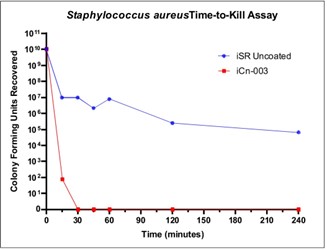
A 2cm2 uncoated PET coupon and a 2cm2 coated sample, iCn003, were inoculated with 1010 CFUs of Staphylococcus aureus for various periods of time. Using the same method as before, the removal of all bacteria cells using a stomacher, dilution and plating out onto suitable agar plates was carried out and the total CFUs enumerated. The results are depicted in Fig. 4. Sample iCn-003 resulted in a LOG 5 reduction or 99.999% reduction after just 15 minutes relative to the control and a complete kill of all Staphylococcus aureus cells after 30 minutes.
Further samples were produced in the industrial batch coater to ensure the process was fully reproducible. The samples were tested in the laboratories at LSTM and, once again, yielded the same results. The five samples tested (iCn-009 to iCn013) all resulted in complete cell death when inoculated with Staphylococcus aureus and incubated at room temperature for 1 hour.
Secondary testing was carried out on one of the samples, iCn-010, to quantify the efficacy of the film in 30 minutes. The percentage of growth relative to the control was measured between 5 and 30 minutes and measurements were taken in 5-minute intervals.
In just 5 minutes, the iCn-010 thin film killed 52.2% of the Staphylococcus aureus CFUs. Within 10 minutes only 12.7% growth was detected relative to the control and by 15 minutes, the coating had resulted in 100% cell death for the pathogen.
Two samples, iCn-010 & iCn-013, were tested against the remaining ESKAPE(e) pathogens. Table 3 numerically states the recovered colony forming units from the two coated samples and the reference coupon. Both coated samples, iCn-010 & iCn-013, displayed high level of biocidal activity when tested against most of the pathogens with an incubation time of 20 minutes. The only pathogen which demonstrated some level of copper resistance was Enterococcus faecium.
Secondary testing was carried out with Enterococcus faecium which displayed only a 47.2% reduction after a 1-hour incubation.
We have been through previous testing with all the ESKAPE pathogens, that the iC-nano coating did in fact completely eradicate Enterococcus faecium when incubated for 2 hours. These results draw the conclusion that Enterococcus faecium requires a longer contact time with the surface for the coating to be effective.
Mechanical Characterization
Throughout development it was central to maintain the biocidal activity of the coatings as well as enhance the mechanical properties of the surface, most importantly the wear resistance of the handles. Copper is widely regarded as a good antimicrobial agent but can often fail when it comes to mechanical durability.
To calculate the wear resistance, an abrasion tester was utilized to perform a reciprocating test on the sample. The aggressive test allowed to determine the friction coefficient of the material. A 6mm diameter tungsten carbide cobalt alloy ball was held within a pin holder. A mass of 50g was placed on top of the pin and remained constant throughout the test. The sample to be tested was fixed in place on the stage and the pin lowered so that it made contact and sat freely upon the top surface of the sample. To simulate 40 years of wear through touch, 800 cycles were programmed in. This was equivalent to 20 minutes total abrasion time, and once started, the pin and WC/Co ball moved linearly across the sample for the duration of the test. The number of cycles were chosen based on an estimated number of uses of the handle over a set period of time.
The software for the apparatus measured the resistive force of friction, Fr, from which the friction coefficient, µ, of the coating could be calculated using the equation below.

The first sample measured had a coating thickness of 40nm which was deposited on a brushed aluminium substrate. The coating failed after ~120 cycles with a coefficient of friction of 0.24. The second sample had a coating thickness of 80nm deposited in the same way on a brushed aluminium substrate. Sample 2 failed after ~400 cycles with a coefficient of friction of 0.2. Once the layer had failed the coefficient of friction of the aluminium substrate measured 1.7 for both Sample 1 and Sample 2. The wear resistive properties of the film can be inferred from a lower coefficient of friction. For these samples, both coefficients were similar but the key takeaway from these tests is the idea that by doubling the thickness of the layer, the lifetime of the protective coating could be almost tripled. The results proved that the thin iC-nano coating provides a protective film for the original substrate, and by increasing the thickness of the layer it is possible to increase the wear resistive properties of the surface in a non-linear manner.
Discussion
Copper is widely accepted as an antimicrobial material. However, when sputtered in its metallic form the thin film is not transparent and lacks the required mechanical properties for touch surfaces. For the visual display application, it is vital that the coatings are completely transparent. For handles and push plates, the coating needs enhanced mechanical properties to withstand wear. These were key factors to consider when developing the iC-nano coating detailed in this paper.
Magnetron sputtering is an ideal choice when it comes to applying antimicrobial technology to a surface or substrate. By sputtering the antimicrobial material onto the surface, it provides a flexible manufacturing process, which when paired with reactive feedback control can create the nano textured films with enhanced mechanical properties which have been presented here. The reciprocating test proved that the wear resistive properties of the film are high and can be improved by increasing the thickness of the layer.
The antimicrobial results provided evidence that the coatings can be instrumental when it comes to combating the ESKAPE(e) pathogens on touch surfaces throughout a hospital setting.
Through rigorous testing at LSTM, the surfaces proved to be effective against the ESKAPE(e) pathogens, resulting in complete cell death in as little as 10 minutes. The nano structure is found to have enhanced the oxidative electrocatalysis of the microorganisms, without compromising the optical properties of the film. Throughout the testing phase, one pathogen, Enterococcus faecium, demonstrated some copper resistance when incubated for shorter time periods (< 2 hours). This proved that the iC-nano coating does not behave the same way for all the ESKAPE(e) pathogens. Further work will be carried out to test the effectiveness of the coatings over time and the associated antimicrobial resistance of the pathogens.
Conclusions
HAIs are a leading cause of death worldwide and with the need to reduce the spread of infection from surfaces, the development of biocidal, self-sanitizing touch surface protection could revolutionize surface cleanliness in healthcare environments. The project has demonstrated that nano-structured copper-based layers offer the highest level of biocidal activity possible based upon standard testing methods.
Gencoa have developed very thin, mechanically enhanced, antimicrobial surface coatings which can be applied to 3D parts or flexible adhesive films. These new copper based thin film coatings have been proven to kill 99.9999% (log 6) of the ESKAPE(e) pathogens in both light and dark environments as quickly as 10 minutes. The layers are solid state and hard wearing, and could last over 10 years based on abrasions tests and simulation.
A R2R production is currently being explored which would allow for a larger throughput of material to be produced at a low cost. The coatings performed well throughout the initial testing and first phase of real-world application. The surfaces are currently installed throughout the Royal Liverpool University Hospital and closely monitored to quantify the biocidal activity and reduction of transmission of disease. Given the efficacy to kill the ESKAPE(e) pathogens, the developed iC-nano coatings should be able to reduce the spread of other pathogens and, when applied in healthcare and other public settings, decrease the incidence of other known infections when applied to suitable common touch surfaces.
IES Technical Sales is a partner of Gencoa. To start a project with us, contact us today.